In the 1860s, Karl Marx wrote that capitalism “produces conditions that provoke an irreparable rift in the interdependent process of social metabolism, a metabolism prescribed by the natural laws of life itself.”1 By preventing the return of essential elements to the soil, capitalist agriculture was breaking a fundamental metabolic cycle.
When Marx was writing, the science of metabolic cycles was in its infancy: an enormous amount has been learned since then, especially in recent decades. To be true to Marx’s method, ecosocialists must, as he did, base our understanding of nature on the best contemporary scientific work.
In essays to be published over the next six months or so, I will attempt to provide a non-technical overview of the relevant science, and discuss how it can contribute to an ecosocialist program for our time. I look forward to your comments, corrections and suggestions.
To begin: what metabolism is, and how it created our world?
Life is the mode of existence of protein bodies, the essential element of which consists in continual metabolic interchange with the natural environment outside them.
—Frederick Engels2
There’s an old joke about a gardener who refused to use a new hoe: “The hoe I’m using is the best I’ve ever had,” he said. “I’ve replaced the handle three times and the head twice.” Organisms, like that hoe, are continuous wholes composed of discontinuous parts. In Steven Rose’s words, “Our bodies are in continuous flux. Nothing about us as organisms is permanent.”
We are built out of tissues arranged into organs, each tissue a mass of individual cells, each cell an assemblage of molecules. We may expect to live for seventy, eighty, ninety years or more. During that time every cell in our body (with the exception of the nerve cells – neurons – in our brain) will have died and been replaced many hundreds or thousands of times. And every one of the giant macromolecules — the proteins, nucleic acids and lipids — of which the cells (including neurons) are constructed will have been laboriously synthesized, and persisted for a few hours, days or months, only to be broken down again and replaced by a successor molecule, a more or less exact copy.3
The average lifetime of a protein molecule in the body of a mammal is around a fortnight. In an adult human, proteins constitute some 10 per cent of body weight, so some 24 grams of protein are being broken down and a fresh 24 grams synthesized every hour of every day — half a gram, or more than a billion billion molecules of protein a minute, throughout our adult life.4
As Lynn Margulis and Dorion Sagan write, this means that living organisms must constantly engage in self-maintenance:
Organisms are far superior to human-built machines. Unlike James Watts’s steam engine, for example, the body concentrates order. It continuously self-repairs. Every five days you get a new stomach lining. You get a new liver every two months. Your skin replaces itself every six weeks. Every year, ninety-eight percent of the atoms of your body are replaced. This nonstop chemical replacement, metabolism, is a sure sign of life. This ‘machine’ demands continual input of chemical energy and materials (food)…
Within the lifetime of a cell, each of five thousand or so different proteins will completely interchange with the surroundings thousands of times. … This energetic maintenance of unity while components are continuously or intermittently rearranged, destroyed and rebuilt, broken and repaired is metabolism.5
Right now, there are trillions of chemical reactions taking place simultaneously in your body. That’s only possible because you, like every other organism from bacteria to great blue whales and giant sequoia trees, draw energy and matter from the world outside your body, and process them in biological factories that no human-made system can emulate. The microscopically tiny cells that make up every living thing are the sites of extraordinarily sophisticated and complex metabolic processes that emerged and evolved over hundreds of millions of years.
When life first appeared on Earth there was no free oxygen — the atmosphere was mostly nitrogen, carbon dioxide and methane. The Sun was dimmer than it is now, but greenhouse gases, in concentrations 10 times higher than today, kept Earth’s surface temperature above freezing, so liquid oceans existed. More heat rose from the planet’s core, driving immense tectonic processes that moved the continents about and caused massive volcanoes that spewed molten rock onto the surface and hot gases into the atmosphere. Because there was no oxygen, there was no protective ozone in the stratosphere, so the surface was constantly bathed in deadly ultraviolet radiation.
There was no biosphere, because there was no life. The emergence of living matter was the first of five great transformations that created the biosphere and changed the Earth.
 Revolution 1: Life
Revolution 1: Life
In 1871, Charles Darwin speculated that life began in “some warm little pond with all sorts of ammonia and phosphoric salts, light, heat, electricity etcetera present.”6 In the 1920s two socialists, working independently, extended Darwin’s thought. The Russian chemist A.I. Oparin and the British geneticist J.B.S. Haldane argued that life must have originated spontaneously in the early ocean, when lightning and solar power triggered the formation of complex organic molecules in that “prebiotic soup.” They held that eventually those organic molecules would combine as living cells, and that life itself had since so changed conditions on Earth that life could not form spontaneously again.
The Haldane-Oparin hypothesis has held up well in broad outline, but recent research has revised most of the details. In particular, we now know that the composition of the atmosphere was very different from what they believed, and that ultraviolet radiation would have killed any living thing that was exposed to the sun. It seems most likely that life originated in or near hydrothermal vents deep in the ocean, where seawater is drawn deep into the ocean floor, is heated and returns containing essential elements of life, including carbon, nitrogen, and sulfur — the “prebiotic soup” in different form. Laboratory experiments have demonstrated that amino acids and other basic building blocks of life form spontaneously in such conditions.
Stephen Jay Gould, who otherwise argued strongly that chance and contingency play a major role in evolution, thought it was significant that that life emerged so early in Earth’s history.
We can only infer from this rapidity that it is not ‘difficult’ for life of bacterial grade to evolve on planets with appropriate conditions. The origin of life may be a virtually automatic consequence of carbon chemistry and the physics of self-organizing systems — given favorable conditions and the requisite inorganic constituents.7
The first life emerged sometime after 4.0 billion years ago, but we may never know exactly when and how, or what the earliest cells were like, because geological processes have destroyed any traces they may have left. The first surviving records are about 3.5 billion years old, and by then life was definitely flourishing. By that time (and probably much earlier) each of the new living things was a microscopic cell with a semi-permeable skin that separated it from its environment, a genetic system (RNA or DNA) that stored genetic information so it could reproduce itself, and a metabolic system that obtained or produced the energy and matter necessary for survival. All of them were prokaryotes, single-celled organisms with no nucleus. They are usually called bacteria, although genetic evidence shows that there are two distinct lineages, bacteria and archaea.8
The construction of the biosphere was underway.
Revolution 2: Metabolic evolution
As biologist Tyler Volk writes, unlike anything that existed before, the new living cells required constant “inputs of nutrients and energy as well as waste outputs. These inputs and outputs are revolutionary new relations…. the cell uses its imports and exports for the molecular manufacturing of its thousands to tens of thousands of special types of molecules.” Just by living, they changed their environments by removing some chemicals and excreting others. The “titanic consequence” was the emergence of biological evolution as cells were forced to adapt or die in constantly changing conditions.9 The existence of life led inexorably to new forms of life.
Bacteria evolve not only by passing mutated DNA to their descendants, but also by directly sharing genes with their neighbors. This “horizontal transfer” allows traits to spread quickly in a local population: combined with the pressure of metabolism-driven environmental change, it enabled rapid biological evolution. While the sizes and shapes of bacteria changed little, their metabolic processes evolved in many directions. Biochemist Nick Lane describes the result as “a cauldron of bacteria, displaying an inventiveness of biochemistry we can only wonder at.”10
Using the basic building blocks of life — carbon, hydrogen, oxygen, nitrogen, phosphorus and sulfur — single-celled bacteria created complex new molecules that enabled them to survive and prosper in many different environments. By 3.2 billion years ago, “bacteria had invented most forms of metabolism, including multiple forms of respiration and photosynthesis.”11
Revolution 3: Photosynthesis
Only through the workings of the most innovating bacteria of all time did the originally anoxic Earth gain an oxygen-rich atmosphere. … It was bacteria that removed the carbon dioxide and produced the oxygen. Bacteria, truly, have made the planetary environment what it is today.12
Photosynthesis has been called “the most important single metabolic innovation in the history of life on the planet.”13
The first living organisms obtained energy from chemical reactions in the sea around them. That worked, but the supply was limited and unreliable: if a vent’s output declined, or water currents shifted, microbes in that area could starve. Those that learned to use solar power were freed from energy scarcity: they multiplied and spread across the planet.
A major metabolic challenge that faced early organisms was obtaining the particular elements they needed to assemble organic molecules, especially carbon, oxygen and hydrogen. The first two were readily available as carbon dioxide, but even near hydrothermal vents the supply of free hydrogen was limited. The first microbes to use solar power used photons to split molecules such as hydrogen sulfide (H2S) into their component elements — in that case, hydrogen and sulfur. The hydrogen was combined with CO2 to make sugars, and the sulfur was discarded or used elsewhere. That method, and others like it, may have been used by some bacteria as early as 3.5 billion years ago.
But an even bigger change occurred later, sometime between 2.9 and 2.5 billion years ago, when a group called the cyanobacteria (often inaccurately called blue-green algae), evolved the ability to capture photons from higher-energy parts of the solar spectrum, and used them to split water into hydrogen and oxygen. The new technology worked anywhere there was water and light, which was just about everywhere. More important, as we’ll see, it led to another revolution: Donald Canfield estimates that “in the end, the use of water in photosynthesis resulted in an increase in rates of primary production [the formation of organic matter] on Earth by probably somewhere between a factor of ten to a thousand.”14
With a few exceptions, all organisms today either use light and photosynthesis to power their metabolisms, or get their energy by eating organisms that do so. The giant molecule that ancient bacteria created to perform the first steps of photosynthesis — ribulose-1,5-bisphosphate-carboxylase/oxygenase, conveniently abbreviated as Rubisco — is still playing that role today and is still the most abundant protein on Earth.
Revolution 4: Oxygen
Photosynthesis gave cyanobacteria the hydrogen and energy they needed to grow and spread. The leftover oxygen changed the world.
Oxygen is a very reactive gas: given the chance, it will combine with (oxidize) a large variety of other elements — to the extent that over 99% of all Earth’s oxygen is in mineral compounds, not in the atmosphere. The first oxygen produced by cyanobacteria was removed in various chemical reactions, so the atmosphere remained oxygen-free for many years, until a tipping point arrived. Scientists disagree on the cause of the transition: some believe that the supply of oxygen-consuming chemicals declined as a result of reduced volcanic activity, but there are other possibilities. What’s certain is that it led, about 2.45 billion years ago, to what’s called the Great Oxygenation Event or the Oxygen Revolution. The waste product of innumerable microscopic organisms began to transform the atmosphere.
The Oxygen Revolution took place in bursts over hundreds of millions of years, but oxygen’s volatility meant that even small amounts had major effects. A layer of ozone formed in the upper atmosphere, blocking deadly ultraviolet radiation — that in turn allowed cyanobacteria to flourish on and near the surfaces of lakes, rivers and oceans, where they produced still more oxygen. Oxygen reactions created two-thirds of the world’s 4500 known minerals — “all of them previously unknown in our Solar System” — and removed dissolved iron from the oceans, depositing bands of iron oxide that contain 90% of the world’s iron ore.15
From this time onward, organisms that use or produce oxygen would dominate biology. Indeed, at the Earth’s surface, only oxygen and carbon dioxide would ever again be abundant enough to supply the needs of cells larger than a few microns, and oxygen would eventually achieve concentrations able to support large, multicellular organisms. From now on, the Earth would start to become our world.16
But before that could happen, the biosphere had to get through a climate crisis.
Metabolic rift
For two billion years the greenhouse gases carbon dioxide and methane had kept Earth’s average temperature above freezing, at levels we would probably find comfortable. Methane’s greenhouse effect is about 86 times greater than carbon dioxide’s, but it doesn’t play a large role in today’s climate because it breaks down in the presence of oxygen. 2.5 billion years ago there was much more than there is today, and it lasted longer, so its role was decisive.
In a relatively short time after the Oxygen Revolution began, chemical reaction with oxygen removed most of the methane in the air. At roughly the same time, the largest land-mass in the northern hemisphere, the super-continent Kenorland, began to break up, exposing large rock faces that absorbed CO2 as they weathered. The greenhouse effect weakened, and the Earth’s average temperature plummeted by an estimated 20°C, to below the freezing point.
The ensuing Huronian Glaciation (named after Lake Huron, where rocks from that period are well-preserved) was the first, longest and most severe of Earth’s ice ages. In three phases between 2.4 and 2.1 billion years ago, kilometer-thick ice spread over land and sea. The combination of oxygen and ice undoubtedly killed many organisms, to the point where some researchers call it the first mass extinction, although there is no hard evidence of that. For some time the entire planet seems to have been covered in ice, creating a Snowball Earth — or Slushball Earth, according to scientists who think the glaciation was less than total. Either way it was, as the authors of a study of that period say, “a climate disaster triggered by the evolution of oxygenic photosynthesis.”17
The term “metabolic rift” usually refers to human interference in or disruption of vital natural cycles, but it clearly applies in this case, when the disruptors were bacteria. The Oxygen Revolution caused a permanent break in the planet’s metabolism, dividing the pre-oxygen past from what geologists call the Proterozoic — Greek for “earlier life” — the time when the predecessors of multi-celled plants and animals developed in a new and different environment.
Revolution 5: New life
Eventually the glaciers retreated, and there wasn’t another ice age for 1.5 billion years. The post-glacial world wasn’t just warmer, it was the setting for another metabolic revolution.
There was a lot more energy to go around in the post-oxidation world, because respiration of organic matter with oxygen yields an order of magnitude more energy than breaking food down anaerobically. Amongst the first organisms to take advantage of this energy source were the first eukaroyotes — complex cells with a nucleus and many other distinct components.18
Bacteria and archaea have survived for nearly four billion years, but their basic physical structure has limited their growth and complexity. Evolving into larger or more complex organisms would require more energy, and their structure imposes physical limits on how much they can acquire. The oxygen that cyanobacteria produced made more energy available, but they and their relatives could not use it efficiently.
We will never know how many failed biological experiments took place in response to the oxygen revolution and the first ice age, when the conditions of life changed permanently, but about 2 billion years ago natural selection produced a major success, the eukaryotes.
Like bacteria, they were single-celled, but they were several orders of magnitude larger, averaging 15,000 times bigger by volume. Their DNA was enclosed in a nucleus, a structure no bacteria ever developed. Instead of generating energy at their outer surfaces, they did so in the semi-independent organs we call mitochondria. Indeed they contained so many new parts and features that Nick Lane describes them as “crammed with assorted jumble.”
Hundreds or even thousands of tiny specialized organs, called organelles, such as mitochondria (in virtually all eukaryotes) and chloroplasts (in algae and plants), mixed up with small sacs of membrane, stacks of folded membranes and a protein skeleton. This riot of compartmentalization gives eukaryotic cells the look of evolution by conglomeration, which is indeed what happened.19

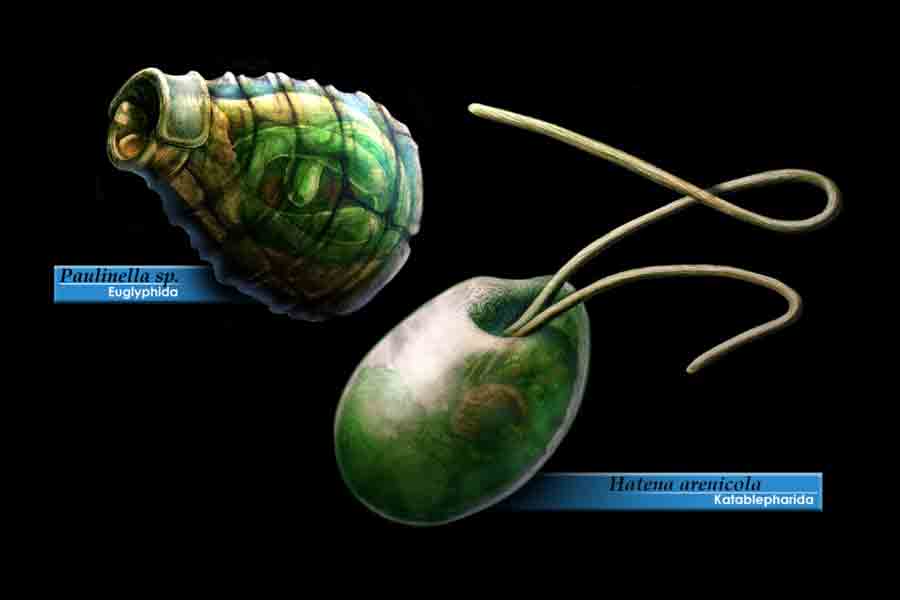
Two examples of “algal” organism, witch independently invented endosymbiosis: Paulinella, a rhizarian amoeba with a cyanobacterium serving as an endosymbiont Hatena, a katablepharid flagellate with a green alga serving as an endosymbiont. Illustration by PavelRiha.
Margulis and Sagan call the new cells “bacterial confederacies … [that] cooperated and centralized, and in doing so formed a new kind of cellular government.”20 Symbiosis, different organisms living cooperatively, isn’t unusual, but this was endosymbiosis — a bacteria, probably a member of the alphaproteobacteria family, took up residence inside another single-celled organism, probably one of the archaea, and the benefits to each were great enough that a permanent division of labor was established. The host provided nutrients for the guest, and the guest provided sugars and energy. If more energy was required, the guest could multiply, enabling increases in size and complexity that could never occur in bacteria. Over time, the host’s descendants acquired most of the guests’ DNA, and the guests evolved into mitochondria, the energy factories that all plants and animals still depend on. Later, cyanobacteria joined the confederacy and evolved into the chloroplasts that plants use in photosynthesis.
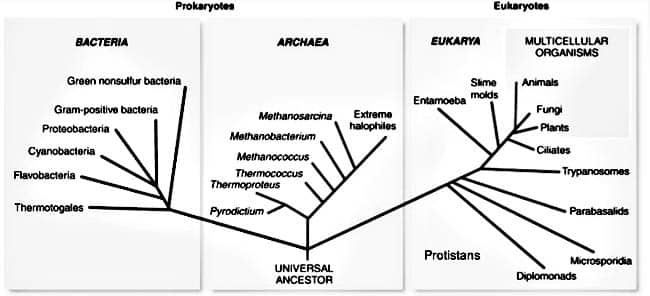
The three domains of life. Prokaryotes emerged about four billion years ago and still exist today. All animals, plants and fungi are descended from the Eucaryotes, which emerged 2 billion years ago.
Eukaryotes were not just a new species, but an entirely new domain of life, a qualitative leap beyond bacteria and archaea. “The void between bacterial and eukaryotic cells is greater than any other in biology,” Lane writes. The new design set the stage for much-accelerated evolution: “In the space of just a few hundred million years — a fraction of the time available to bacteria — [eukaryotes] gave rise to the great fountain of life we see all around us.”21 All multi-celled organisms, all animals, plants and fungi, are descended from the revolutionary confederacies that emerged two billion years ago, in the wake of the first ice age and the Oxygen Revolution.
Life, Metabolism, Biosphere
Earth System scientists now think in terms of the coupled evolution of life and the planet, recognizing that the evolution of life has shaped the planet, changes in the planetary environment have shaped life, and together they can be viewed as one process.22
I started this project after reading one skeptic who dismissed metabolic rift as merely a metaphor, and another who claimed that Marx was wrong because metabolism is just chemical processes inside your body. The quotation from Engels at the beginning of this essay replies to both. Metabolism is a fundamental feature of all life, and it always involves exchanges with the world outside the organism, exchanges that are intimately connected to the biogeochemical cycles that shape and maintain the entire Earth System.
Life cannot exist without metabolism, and metabolism cannot exist without changing the world. Grasping that principle is an essential first step towards understanding the relationship between society and the rest of nature, and the causes of today’s ecological crisis.
The first cells to emerge on the barren planet immediately began changing the world, and were themselves changed in the process. There was no plan and no designer: the five revolutions that created and shaped the biosphere were results of millions of years of physical, chemical and biological experiments, filtered by natural selection.
The history of life on Earth, and thus of humanity, is ultimately the history of the changing ways in which living things have obtained, processed and exchanged matter and energy. Four billion years of co-evolution have produced innumerable species and life-webs that are metabolically connected at every level, from the smallest cell to the entire planet.
Future articles will discuss those connections, and consider how capitalism is by disrupting them and threatening Earth’s life earth support systems.
References
- ↩ Karl Marx, Capital Volume 3 (Penguin: 1981 [1894]), 949.
- ↩ Frederick Engels, Dialectics of Nature (International Publishers: 1940), 195-7.
- ↩ Steven Rose, Lifelines: Life Beyond the Gene (Oxford University Press: 1997), 38.
- ↩ Ibid, 158.
- ↩ Lynn Margulis and Dorion Sagan, What is Life? (Simon & Schuster: 1995) 23-4.
- ↩ Charles Darwin to Joseph D. Hooker, February 1, 1871.
- ↩ Stephen Jay Gould, “Life on Mars? So What?” New York Times, August 11, 1996.
- ↩ The differences are in their DNA and chemical composition. Many archaea are extremophiles, thriving in conditions that would be deadly for other organisms.
- ↩ Tyler Volk. Quarks to Culture: How We Came to Be (Columbia University Press: 2017), 73, 75.
- ↩ Nick Lane, The Vital Question, 29.
- ↩ Ibid.
- ↩ Margulis and Sagan, What is Life?, 69, 71.
- ↩ Margulis and Sagan, Microcosmos, 78.
- ↩ Donald E. Canfield, Oxygen: A Four Billion Year History (Princeton University Press: 2014), 42.
- ↩ Robert M. Hazen, The Story of Earth, (Viking Penguin: 2012), 168, 151.
- ↩ Andrew H, Knoll, Life on a Young Planet (Princeton University Press: 2015), 107.
- ↩ Robert Kopp, Joseph Kirschvink, Isaac Hilburn, and Cody Nash, “The Paleoproterozoic snowball Earth,” Proceedings of the National Academy of Sciences, Vol 102, No 32, August 2005.
- ↩ Tim Lenton, Earth System Science: A Very Short Introduction (Oxford University Press: 2016), 69.
- ↩ Nick Lane, Oxygen: The Molecule that Made the World (Oxford University Press: 2002), 150.
- ↩ Margulis and Sagan, Microcosmos, 117.
- ↩ Nick Lane, Power Sex, Suicide: Mitochondria and the Meaning of Life, (Oxford University Press: 2005), 27.
- ↩ Tim Lenton, Earth System Science, 57.