In reporting on the nuclear situation in Japan, terms describing the possible state of the reactors have been used by Japanese officials and international experts. Unfortunately, these terms are not always used consistently. We discuss some of them below.
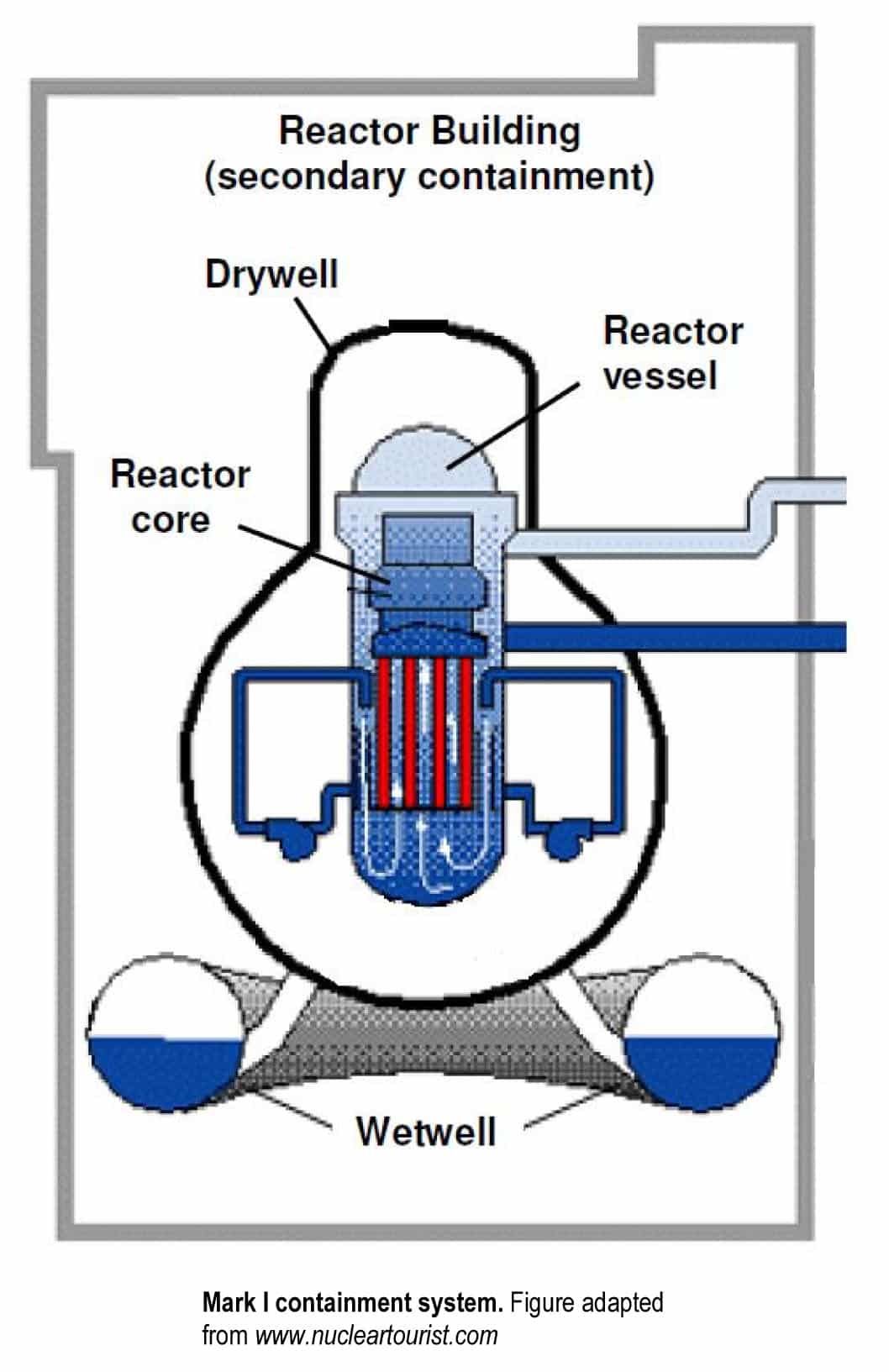 |
The reactor core containing nuclear fuel is the “engine” of a nuclear power reactor. The reactor core resides in the lower region of a metal “pot” called the reactor vessel. The reactor vessel is enclosed within the primary containment structure, consisting of the drywell and wetwell (see diagram), which is designed to contain radioactive materials released during a reactor accident. The reactor building provides the secondary containment, which is intended to prevent any leaks from the primary containment from escaping to the environment. The air in the reactor building is sent through filters to remove any radiation before being released to the outside.
The fuel consists of uranium or uranium/plutonium pellets stacked inside long tubes made from zirconium metal. When a reactor is operating, the fuel gets very hot. The fuel is immersed in water, and the heat produces steam, which is used to drive a turbine to produce electricity. The water also keeps the fuel from overheating, and is continuously circulated through the reactor core to carry away excess heat. Even if the reactor shuts down, the fuel will remain hot for a long time, so must still be cooled.
Fuel Damage
If the pumps that circulate the cooling water are not operating, the water in the reactor vessel will heat up and evaporate, and the fuel can become uncovered inside the reactor vessel. This situation also occurs if something like a pipe rupture causes the water to drain out of the reactor vessel. At this point, the zirconium cladding on the fuel rods will start to heat up, blister, and then rupture. Radioactive material that normally collects in the gap between the fuel pellets and the cladding is then released into the reactor vessel.
Radioactivity Release Pathways
The radioactive material released from damaged fuel into the reactor vessel can get into the primary containment. To protect the reactor vessel and attached piping from rupturing due to high pressure, relief valves automatically open to discharge steam — and the radioactive material along with it — into the primary containment structure. Workers may also manually open the relief valves to prevent high pressure in the reactor vessel from impeding the flow of makeup water, such as the sea water that has reportedly been injected into some of the Japanese reactors.
In addition, a steam-driven emergency system called the reactor core isolation cooling (RCIC) system uses steam from the reactor vessel to spin a small turbine connected to a pump that transfers makeup water to the reactor vessel. After the steam is used for this purpose, the steam — along with the radioactive material — is deposited into the primary containment.
Partial Meltdown
If the fuel is uncovered by water and exposed for a period of hours, it will start to melt. This makes cooling more difficult as the melted fuel clogs the spaces between the fuel rods. The melted fuel will start to collect on the bottom of the steel reactor vessel (sometimes called the lower head). The molten fuel will begin to burn its way through the reactor vessel.
Complete Meltdown
If the water level is low enough, essentially all the fuel in the core can melt and will fall to the bottom of the reactor vessel. It will be a matter of hours before the fuel melts through the steel reactor vessel and onto the concrete floor of the primary containment. The containment is designed to contain the melted fuel and its radioactive emissions, but there are ways in which the primary containment can fail in the event of a meltdown; this has been a concern with the Mark I containment, which the affected Japanese reactors use. The emissions will generate increasing pressure over a period of days and weeks, which can lead to collapse of the primary containment if it is not relieved.
The primary containment has a typical leak rate of roughly 1% of its volume per day, so some of the radioactive material will leak into the secondary containment (the reactor building). The reactor building is normally kept at a reduced pressure relative to the outside so any radioactive emissions will stay inside the building. However, this requires the building itself to be intact.
A meltdown does not necessarily mean that there will be a large release of radioactivity. This will depend on the integrity of the primary and secondary containments.
Radioactive Isotopes
Radioactive materials decay, releasing particles that can damage living tissue and lead to cancer. Some elements have different forms, called isotopes, that differ in the number of neutrons in the nucleus. The radioactive isotopes of greatest concern in a nuclear power accident are iodine-131 and cesium-137.
Iodine-131 has a half-life of 8 days, meaning half of it will have decayed after 8 days, and half of that in another 8 days, etc. Therefore, it is of greatest concern in the days and weeks following an accident. It is also volatile so will spread easily. In the human body, iodine is taken up by the thyroid, and becomes concentrated there, where it can lead to thyroid cancer in later life. Children who are exposed to iodine-131 are more likely than adults to get cancer later in life. To guard against the absorption of iodione-131, people can proactively take potassium iodine pills so the thyroid becomes saturated with non-radioactive iodine and is not able to absorb any iodine-131
Cesium-137 has a half-life of about 30 years, so will take more than a century to decay by a significant amount. Living organisms treat cesium-137 as if it was potassium, and it becomes part of the fluid electrolytes and is eventually excreted. Cesium-137 is passed up the food chain. It can cause many different types of cancer.
This fact sheet was published by the Union of Concerned Scientists on 13 March 2011; it is reproduced here for non-profit educational purposes.
var idcomments_acct = ‘c90a61ed51fd7b64001f1361a7a71191’;
var idcomments_post_id;
var idcomments_post_url;
