This is an update of my April 7 post on cancers resulting from Chernobyl — this post includes more detailed information and a more recent estimate of the total radioactive dose from the Chernobyl accident.
There is a lot of confusion about how many excess cancer deaths will likely result from the 1986 Chernobyl accident in Ukraine. There are two main sources of confusion: information that is confusing — and in some cases misleading — put out by authoritative sources, and large inherent uncertainties in estimates of the effects of the accident. Because of these inherent uncertainties, it is perhaps most appropriate to only cite order-of-magnitude results: the numbers of excess cancers and cancer deaths worldwide will be in the tens of thousands.
However, based on the data given below, 53,000 and 27,000 are reasonable estimates of the number of excess cancers and cancer deaths that will be attributable to the accident, excluding thyroid cancers. (The 95% confidence levels are 27,000 to 108,000 cancers and 12,000 to 57,000 deaths.) In addition, as of 2005, some 6,000 thyroid cancers and 15 thyroid cancer deaths have been attributed to Chernobyl. That number will grow with time.
Much lower numbers of cancers and deaths are often cited, but these are misleading because they only apply to those populations with the highest radiation exposures and don’t take into account the larger numbers of people who were exposed to less radiation.
One authoritative but misleading report on the consequences of Chernobyl is Chernobyl’s Legacy: Health, Environmental and Socio-economic Impacts, released by the UN-sponsored Chernobyl Forum (September 5, 2005). According to this report (p. 15):
The international expert group predicts that among the 600 000 persons receiving more significant exposures (liquidators working in 1986-87, evacuees, and residents of the most “contaminated” areas), the possible increase in cancer mortality due to this radiation exposure might be up to a few percent. This might eventually represent up to four thousand fatal cancers in addition to the approximately 100 000 fatal cancers to be expected due to all other causes in this population. Among the 5 million persons residing in other “contaminated” areas, the doses are much lower and any projected increases are more speculative, but are expected to make a difference of less than one per cent in cancer mortality.
(The “most contaminated” areas were those with contamination greater than 555 kilobecquerel per square meter [kBq/m2] of Cesium 137 [Cs-137], and the “contaminated” areas had greater than 37 kBq/m2 of Cs-137.)
Roughly 20% of people die of cancer, so we would expect a million fatal cancers in a population of 5 million. A one percent increase would translate into 10,000 additional cancer deaths, so according to this report, there would be fewer than 10,000 excess cancer deaths among those in the contaminated areas. These deaths would be in addition to the 4,000 fatal cancers among evacuees, liquidators (clean-up workers), and those in the most contaminated areas.
Another UN Chernobyl Forum report from the following year, Health Effects of the Chernobyl Accident and Special Health Care Programmes, gives a total of 5,000 for the excess cancer deaths “predicted” for the inhabitants of the contaminated areas. This is consistent with the 2005 report, as it represents about a 0.5% increase in cancer mortality.
Because of the 2005 report, people frequently cite “4,000” as the number of eventual excess cancer fatalities. However, by limiting its analysis to people with the greatest exposure to released radiation, the report seriously underestimates the number of cancers and cancer deaths attributable to Chernobyl. The effects of the radiation were not limited to the “contaminated” areas but would be felt in Europe and beyond.
The current understanding of the relationship of cancer to radiation is that the risk of solid cancers increases linearly with dose and that there is no safe amount of radiation. This understanding is represented by the “Linear No-Threshold” (LNT) model of cancer. For leukemia, a blood cancer, a linear-quadratic model is generally used.
We can estimate the number of additional cancer deaths using data from several publications of the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR), which give estimates of the radiation exposure from the accident. Unfortunately, the information is not all available in one publication.
Annex D “Health effects due to radiation from the Chernobyl accident” of the Committee’s 2008 report Sources and effects of ionizing radiation (Appendix B) includes information on the collective effective doses for different populations in Belarus, Ukraine, Russia, and the rest of Europe. (The collective effective dose is the average effective dose multiplied by the number of exposed people. The average effective dose takes into account the different biological effects of different types of radiation on different types of tissue in the body, and a sievert, or Sv, is a measure of the radiation dose that takes into account the effects of different types of radiation.)
Earlier UNSCEAR publications give the collective lifetime dose for these populations. In the 2008 report, the collective effective dose is determined from 1986, when the accident occurred, to 2005. According to the 2008 report, this represents 80% of the lifetime dose.
Moreover, unlike earlier editions of the UNSCEAR reports, the 2008 report does not include information on the dose for populations outside of Europe. By omitting this information, the report erroneously suggests the effects of Chernobyl were limited to Europe. Other areas within the northern hemisphere — including Asia, Africa, and the Americas — were also contaminated by the Chernobyl accident.
Using data from the 1988 UNSCEAR report in Table 24, we find that the collective effective lifetime dose for areas outside of Europe is 64,000 person-Sv. The collective lifetime doses for the USSR (which includes Belarus, Ukraine, and Russia) and the rest of Europe are given as 226,000 and 330,000 person-Sv, respectively. In contrast, the 2008 estimates for the collective lifetime doses for the USSR and the rest of Europe are 156,000 and 163,000 person-Sv. We use the more recent estimates.
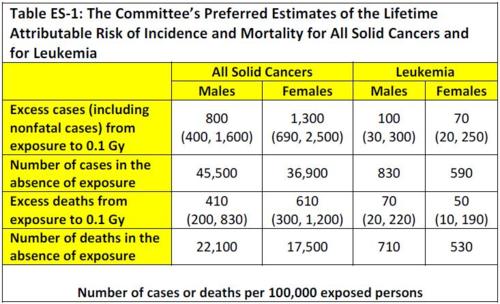
These doses are from low levels of gamma rays emitted by Cs-137, which is a type of low linear energy transfer (LET) ionizing radiation. Accordingly, we can relate the collective effective lifetime dose received in these various areas to the number of excess cancers and cancer deaths using data from Table ES-1 of the 2006 NAS report Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII Phase 2 (Washington, DC, p. 15), which we reproduce below:
The numbers in parentheses are “95% subjective confidence intervals,” which the report states
. . . reflect the most important uncertainty sources — namely, statistical variation, uncertainty in the factor used to adjust risk estimates for exposure at low doses and low dose rates, and uncertainty in the method of transport.
For gamma and beta radiation, which are of interest here, 1 Sv = 1Gray (Gy). Assuming an equal number of males and females, we see that the expected incidence and mortality of solid cancers and leukemia are 0.1135 cancer cases and 0.057 cancer deaths per Sv. The 95% uncertainty intervals are (0.057, 0.2325) for cancer cases and (0.0265, 0.122) for cancer deaths per Sv.
For example, for a collective dose of 465,000 person-Sv, the expected number of cancer cases would be 53,000, of which some 27,000 would result in death. If we apply the lower and upper confidence bounds, we find a range of 27,000 to 108,000 excess cancer cases, of which 12,000 to 57,000 would be fatal.
The estimated collective doses and the health consequences for different populations and areas are shown in the table below. Except where noted, all numbers are from the 2008 UNSCEAR report referenced above. Note that the collective effective doses in column 5 below are lifetime doses and are 25% greater than the doses given in the UNSCEAR report, which are only for the period 1986-2005. Moreover, the collective effective dose excludes the collective thyroid dose, and the excess solid cancers do not include thyroid cancer.
Finally, note that the numbers of cancer deaths predicted by the 2005 and 2006 Chernobyl Forum reports — 4,000 for the recovery operation workers, evacuees, and residents of the more contaminated areas, and 5,000 for the remaining residents in the contaminated areas — are consistent with the values of 5,300 and 3,000 we calculate below.
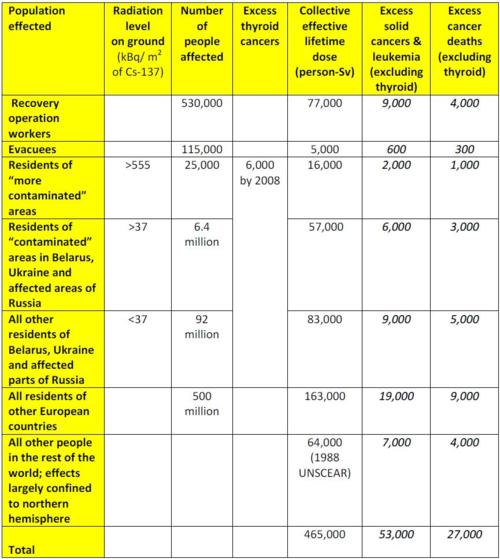
Table 2: Numbers in italics are calculated.
Note that because exposure only increases the probability of developing cancer, in general no given cancer can be attributed to Chernobyl. Moreover, because these additional cancers will be distributed among hundreds of millions of people, they will not be discernable among all the other cancer cases. (Table ES-1 indicates that, on average, about 42% of people have cancer at some point in their lives, and about 20% of people die of cancer.)
In contrast, epidemiological studies have found that the large increase in thyroid cancers among children in Belarus, Ukraine, and Russia following the accident clearly indicates that it was the cause of the increase. According to the 2008 UNSCEAR report (p. 19):
To date, some 6,000 thyroid cancers have been seen among those in the three republics who were under 18 at the time of the accident, of which a substantial fraction is likely to have been due to radiation exposure.
A 2006 study that also uses a linear no-threshold model finds comparable estimates of the numbers of cancers and cancer deaths. “Estimates of the Cancer Burden in Europe from Radioactive Fallout from the Chernobyl Accident” by E. Cardis et al. in the International Journal of Cancer concludes that Chernobyl will have caused 16,000 thyroid cancers and 25,000 other cancers in Europe by 2065, and that 16,000 of these cancers will be fatal. Since thyroid cancer is rarely fatal, most of the cancer deaths will be from other cancers. (These estimates do not consider the recovery-operation workers.)
The numbers of lifetime cancers caused would be slightly higher than the Cardiset al. figures above: at 17,000 thyroid cancers, 27,000 other cancers, and 17,000 fatal cancers. (According to Cardis et al., the dose from 1986 to 2005 is 85% of that from 1986 to 2065. As noted above, the dose to 2005 is 80% of the lifetime dose. Thus, the number of lifetime cancers is 85/80 times the number of cancers by 2065.)
We can compare these numbers with the ones calculated above using the UNSCEAR data. Excluding the recovery workers, the figures above yield 36,000 other cancers and 18,000 cancer deaths in Europe, which are comparable to the Cardis et al. figures of 27,000 other cancers and 17,000 cancer deaths.
Finally, we note that a 2009 book by Alexey V. Yablokov, Vassily B. Nesterenko, and Alexey V. Nesterenko, Chernobyl: Consequences of the Catastrophe for People and the Environment, published by the New York Academy of Sciences, concludes that “a more realistic figure is 212,000 to 245,000 [cancer] deaths in Europe and 19,000 in the rest of the world” (p. 161). These values are an order of magnitude greater than those cited above.
The book is based on a wide variety of material, which has been compiled in a manner that is difficult to discern. According to the book (p.xi):
. . . the literature incorporated into the present volume includes about 1,000 titles and reflects more than 5,000 printed and Internet publications, primarily in Slavic languages.
Moreover, the book notes that at least some of this source material would be rejected by “Western” scientists (p.37):
It is correct and justified for the whole of society to analyze the consequences of the largest-scale catastrophe in history and to use the enormous database collected by thousands of experts in the radioactively contaminated territories, despite some data not being in the form of Western scientific protocols. This database must be used because it is impossible to collect other data after the fact.
Given this disclaimer, we have to discount the conclusions of this book, at least unless and until further information becomes available.
Dr. Lisbeth Gronlund is a senior scientist and co-director of the Global Security Program at the Union of Concerned Scientists (UCS) in Cambridge, Massachusetts and a research affiliate in the Program on Science, Technology, and Society at the Massachusetts Institute of Technology (MIT). This article was first published in the UCS’s All Things Nuclear blog on 17 April 2011; it is reproduced here for non-profit educational purposes.
| Print